In a groundbreaking study at the Ulsan National Institute of Science and Technology (UNIST), researchers have unveiled an innovative catalyst that has the potential to transform the way we tackle environmental pollution. Under the leadership of Professor Jaeheung Cho, the team has successfully engineered a catalyst that emulates the functions of natural metalloenzymes. This advancement could signal a significant leap toward eco-friendly and energy-efficient pollution mitigation strategies.
The Science behind the Catalyst
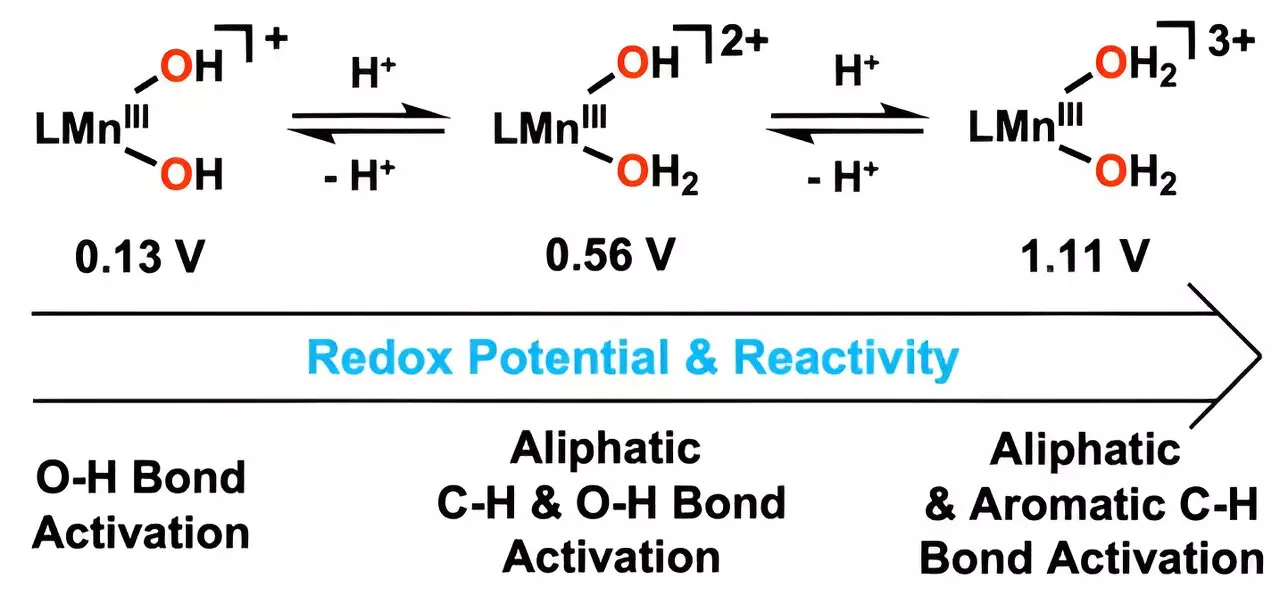
At the heart of this research lies the unique ability of the newly developed catalyst to oxidize harmful hydrocarbons, particularly through the breakdown of stubborn carbon-hydrogen (C–H) bonds. By skillfully introducing hydrogen ions into the hydroxo ligand, the team generated metal-bound water molecules that serve as the catalyst’s active components. The result? A catalyst that demonstrates remarkable efficacy in degrading toxic hydrocarbons at considerably lower temperatures compared to existing methodologies.
Professor Cho has aptly highlighted the significance of this breakthrough: the activated manganese catalyst has shown an impressive capacity to accelerate the oxidation process, fundamentally altering reaction dynamics. The improved reduction potential of manganese—achieved through careful modifications—opens the door for wider applications in sustainable technology.
Lower Temperatures, Greater Effectiveness
What distinguishes this catalyst from its predecessors is its capability to perform complex chemical reactions at milder temperatures, thus conserving energy and minimizing environmental impact. It is a game-changer, especially in industrial applications where energy consumption is a growing concern. The implications are enormous—with industries potentially reducing operational costs while meeting stringent environmental regulations.
The success of this catalyst was demonstrated by the effective oxidation of anthracene, a hydrocarbon known for its resilience and strong C–H bonds. By efficiently breaking these bonds at lower temperatures, the researchers were able to eliminate toxicity, marking a significant milestone in the field of environmental chemistry.
Expanding the Horizon of Catalysis
Additionally, the catalyst excels in decomposing aromatic hydrocarbons, a category notorious for being water-insoluble and chemically resistant. This ability not only underscores the remarkable versatility of the manganese-based catalyst but also suggests far-reaching possibilities for wastewater treatment and soil remediation.
Professor Cho has made a pivotal statement regarding the advancement in manganese chemistry, noting, “This is the first instance where a manganese(III) complex with two water molecules has reacted with aromatic hydrocarbons at low temperatures.” This assertion emphasizes the unprecedented nature of their findings and sets the stage for further exploration into the realm of metal catalysis.
Future Outlook: A Step Toward Sustainable Technologies
As we face increasing challenges from pollution and environmental degradation, the insights gleaned from this research could herald a new era in how we approach chemical reactions in environmentally sensitive contexts. By controlling the reduction potential of manganese, the researchers aim to contribute significantly to the development of next-generation metal catalysts that are not only effective but also sustainable.
The work done by the team at UNIST serves as a reminder of the profound impact basic scientific research can have on global environmental issues. It illustrates how mimicking nature can lead to innovative solutions, pushing the boundaries of what is achievable in catalysis and pollution control.
Leave a Reply