In the realm of organic chemistry, the ability to manipulate molecular structures at a singular atom level is akin to wielding a magic wand. This necessary precision can unravel complex problems, paving the way for more efficient synthesis of intricate pharmaceuticals. Pioneering this endeavor is Professor Max Martin Hansmann and his team from the Department of Chemistry and Chemical Biology, who have introduced a groundbreaking reagent designed for the selective addition of carbon atoms to various molecules. This development, recently published in the prestigious journal Science, signifies not just incremental progress, but a potential paradigm shift in how chemists approach organic synthesis.
The Challenge of Carbon Atom Incorporation
The selective incorporation of carbon atoms has posed significant challenges in the field of organic chemistry, primarily due to the instability associated with carbon sources. Traditional methods often involve toxic reagents that compromise safety and simplify reaction paths. Addressing these critical pain points, Professor Hansmann’s team has devised a reagent that acts both as a reliable source of carbon and a multipurpose transfer agent. Their innovative approach emphasizes scientific elegance—a term frequently invoked by researchers aiming to achieve superior results with minimal complexity.
The Team Behind the Discovery
At the heart of this project is Dr. Taichi Koike, a postdoctoral researcher and the primary author of the study. Currently a recipient of the Alexander von Humboldt Research Fellowship, Koike has been pivotal in crystallizing the practical aspects of their research. The combination of Hansmann’s seasoned experience with Koike’s fresh insights created a fertile environment for scientific advancement—a reminder of how collaboration amplifies potential in the laboratory setting.
The Significance of the New Reagent
The reagent developed by Hansmann and his team marks a substantial advancement in carbon chemistry. It is characterized by its dual capability: acting as both a source of carbon atoms and a facilitator for their transfer. Professor Hansmann expresses optimism regarding the wide-ranging implications of this research, suggesting that the enhancer could serve in various domains such as the creation of higher cumulenes or facilitate late-stage functionalization of complex molecular structures. Such versatility may unlock unforeseen applications and reduce the synthetic sequence needed for crafting complex pharmaceuticals.
A New Approach to Stabilizing Carbon
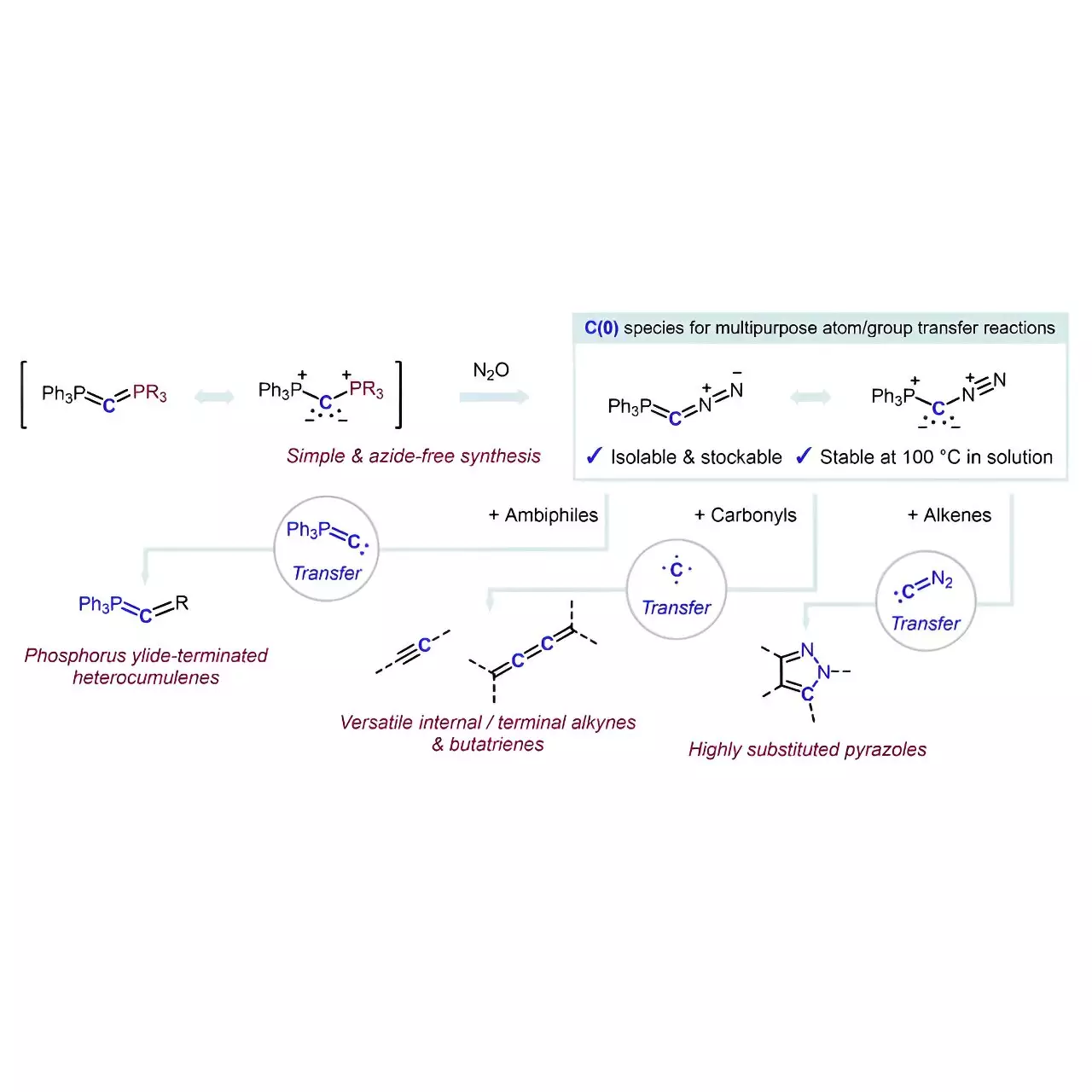
One of the ingenious elements of their reagent lies in its stability, achieved through a straightforward method of coordinating a carbon atom with two neutral electron-donating groups. Dubbed carbones (L1→C←L2), these carbon components had seen only limited exploration in the past as viable carbon atom sources. The conceptual leap of flanking a carbon atom with two labile groups (PPh3 and N2) has set a new precedent for stability without compromising functionality. This innovative stabilization technique not only streamlines the process but significantly enhances safety, a crucial element in chemical synthesis.
A Practical Application: The Synthesis Process
The synthesis of the reagent Ph3P=C=N2 exemplifies the elegance of their approach. Achieved through a formal PPh3/N2 exchange reaction, the process sidesteps the commonly employed azides that frequently pose safety risks. Instead, leveraging carbodiphosphoranes and nitrous oxide, the researchers have executed a pathway that is both straightforward and innovative.
What really stands out is the reagent’s capacity to act as a selective transfer agent. The phosphorus ylide-terminated heterocumulenes derived from ambiphiles and the ability to facilitate reactions with alkenes showcases the potential diversity in applications for this new reagent.
Implications for Future Research
The implications of this research extend well beyond the reactions themselves. As Professor Hansmann and his group continue to explore the reactivity of this newly synthesized reagent, a plethora of applications may arise in the fields of drug discovery and material science. The efficient transfer of carbon atoms could redefine methodologies in organic synthesis, allowing for the rapid development of new medicinal compounds with unprecedented potency and specificity.
The work being conducted by Professor Hansmann and his team not only represents a technical milestone in reagent development but also highlights the creative problem-solving inherent in research. Their commitment to addressing the fundamental challenges in carbon chemistry is opening new vistas in organic synthesis, and it will be exciting to witness the next stage of innovations that will stem from their groundbreaking work.
Leave a Reply