In an era where the climate crisis looms larger than ever, the quest for sustainable energy sources is paramount. Recent advancements in the field of chemistry signal a promising direction in harnessing waste materials to create high-value resources. A landmark study published in *Nature Catalysis* illustrates how innovative methods that integrate electricity and advanced catalysis are offering efficient pathways to convert carbon dioxide, a notorious greenhouse gas, into methanol—a highly desired liquid fuel. This shift not only touches on environmental sustainability but also opens doors to a myriad of applications that could redefine fuel consumption.
The Mechanics of the Reaction
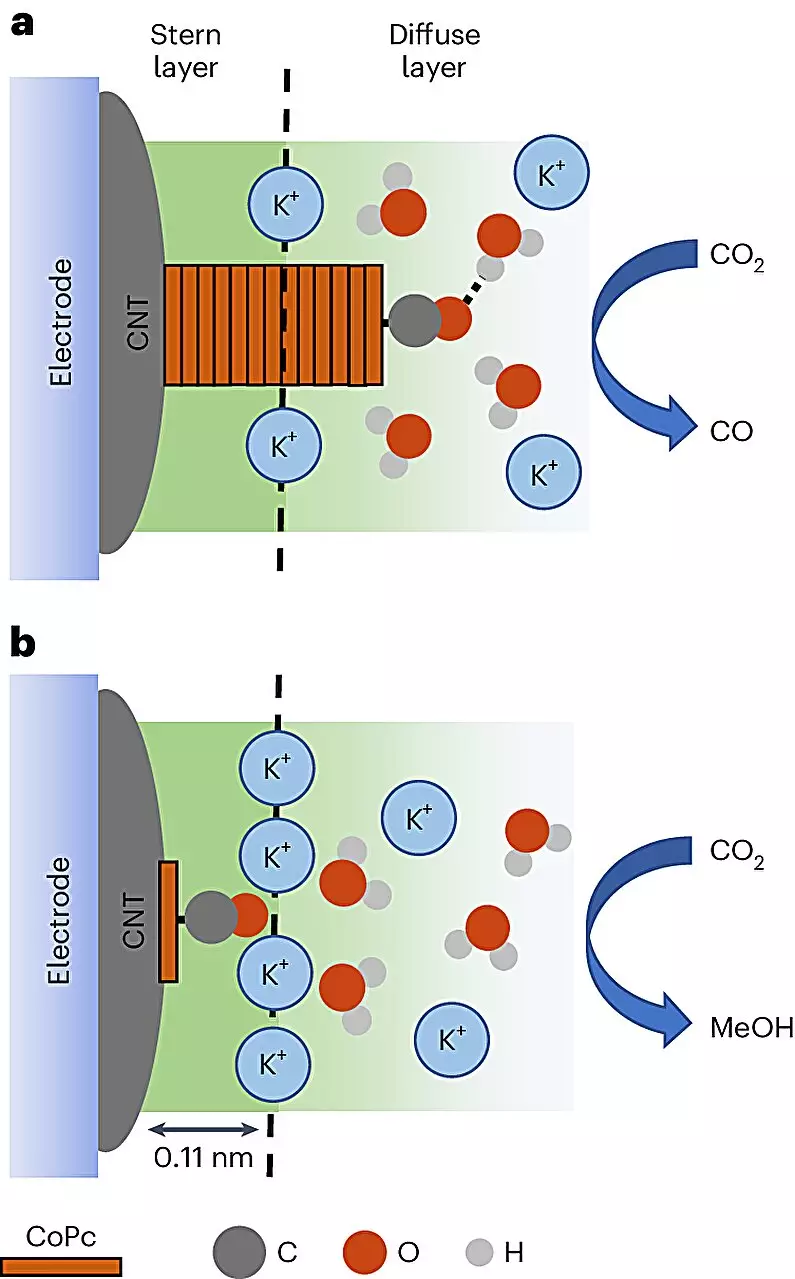
At the core of this groundbreaking research is the utilization of cobalt phthalocyanine (CoPc) molecules, which are catalytically active compounds, applied to the surface of carbon nanotubes. This strategy capitalizes on the unique electrical properties of these graphene-like structures to foster efficient interactions during electrochemical reactions. An electrolyte solution was implemented to facilitate the required electrical current, which instigates the reduction of carbon dioxide into methanol. This representation of chemistry is not just a simple formula; rather, it embodies a carefully choreographed dance on a molecular level, where scientific precision meets innovative engineering.
Utilizing advanced in-situ spectroscopy techniques, researchers could visualize the catalytic environment, ultimately discerning how variations in conditions impact product formation. For the first time, the researchers captured the dynamism of molecular interactions, showcasing transformative moments where carbon dioxide is either successfully converted into methanol or wasted by being turned into less desirable carbon monoxide. This nuanced understanding enables chemists to optimize the catalytic pathways and increase the efficiency of methanol production up to eight times, showcasing a significant breakthrough that has implications far beyond this specific reaction.
The Importance of Reaction Environment
The research underscores the significance of the reaction environment in driving the catalytic process. As co-author Robert Baker poignantly notes, the nature of the catalyst’s distribution on the surface of carbon nanotubes plays a crucial role in steering the reaction toward methanol production. This strategic manipulation provides essential insights into designing more effective catalytic systems. The scientific community’s longstanding challenge has been to comprehend the microscopic interactions dictating catalyst performance—a conundrum this study begins to unravel.
Through this investigation, researchers achieved a deeper understanding of not only the chemical identities involved but also the physical narratives that play out on the molecular stage. An essential aspect is the interaction of CoPc with cations—supercharged particles that augment the methanol generation process. This initial exploration into the realm of cation involvement may lead to further studies clarifying their broader implications and promises in enhancing catalytic performances overall.
A Catalyst for Broader Advancements
The implications of this research extend beyond the laboratory. With methanol being a contender for alternative fuels, the ability to produce it cleanly from carbon dioxide suggests a viable route toward decarbonizing the transportation sector and beyond. Notably, methanol’s high energy density positions it among the most effective energy carriers available today, with applications spanning from fuel for motor vehicles to prospects for heating and electricity generation.
Furthermore, this research represents a decisive movement towards a circular economy, where waste is transformed into fuel rather than contributing to environmental degradation. The potential of harnessing renewable electricity in this context could catalyze revolutionary progress in the development of green technologies, enriching not only energy strategies but also influencing various sectors by providing clean energy sources and fostering chemical innovation.
The Path Forward: Exploring New Horizons
This study instigates an exciting dialog within the scientific community, challenging existing paradigms in catalysis and sustainable chemistry. It lays a compelling foundation for future research endeavors aimed at refining electrochemical processes and enhancing the efficiency of waste-to-fuel conversions. The vibrational signatures captured in this research signify a wealth of uncharted territory, beckoning further exploration into both cation interactions and optimization techniques that could revolutionize how we perceive energy production.
In essence, this intriguing breakthrough invites not only chemists but also policymakers and industries to reconsider their approaches to energy consumption and resource management. By threading together innovative techniques with a commitment to sustainability, the research opens avenues that promise to shape both our environmental landscape and economic future. Through collaborative efforts and unyielding curiosity, the goal of transforming carbon emissions into sustainable fuel may well be within our grasp, ushering in a new era of energy that is both responsible and resilient.
Leave a Reply