At the forefront of materials science, researchers continually grapple with crafting highly efficient catalysts that can operate at the atomic level. A recent study emanating from the University of Science and Technology of China, led by Prof. Yan Wensheng, sheds light on a pivotal challenge in single-atom catalysis: the intricate balance between metal loading and catalytic performance. This exploration is not merely about enhancing efficiency; it is a quest to construct catalysts that push the boundaries of modern technology while considering cost-effectiveness and sustainability.
The Intricacies of Metal Loading in Catalysts
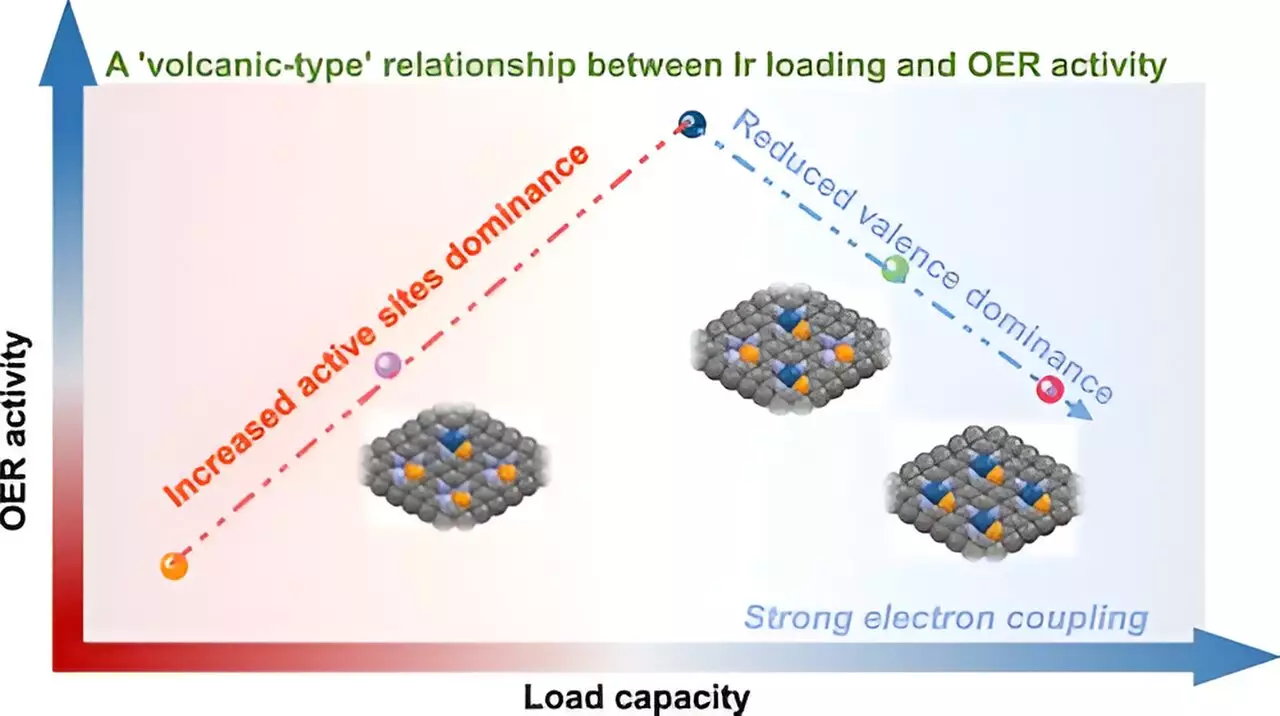
Catalysts play a crucial role in facilitating chemical reactions, particularly in processes such as the acidic oxygen evolution reaction (OER). However, achieving optimal metal loading—where the catalyst operates at its peak without succumbing to aggregation—represents a monumental challenge. The research team’s innovative approach involved a P-anchoring strategy to synthesize iridium (Ir) single-atom catalysts with metal loadings between 5% and 21wt%. This nuanced methodology hinges on devising a delicate balance where increasing metal atoms enhances performance until a critical threshold is surpassed.
The ‘Volcano-Type’ Relationship: A Novel Discovery
The study unveiled a remarkable discovery—a “volcano-type” relationship that underpins the correlation between metal loading and OER activity. This metaphorical depiction illustrates that initially, as Ir atoms are added, catalytic performance ramps up proportionally. However, once a peak is reached, an oversaturation effect plays out: the interaction between closely positioned Ir atoms diminishes the inherent catalytic properties of the catalyst. This nuanced understanding prompts a re-evaluation of loading strategies previously deemed efficient, underscoring the importance of delicate manipulation in catalyst design.
Innovative Techniques Driving Insight
Utilizing advanced techniques such as synchrotron radiation XAS spectroscopy and X-ray photoelectron spectroscopy (XPS), the researchers meticulously identified the Ir-P coordination that serves as the stabilizing agent for the catalyst. Such clarity enhances understanding of electron interactions at an atomic scale, presenting a significant breakthrough in the field. The combined theoretical computations considered alongside experimental data empower other researchers by providing a foundation for the tailored design of future catalysts that can tackle OER efficiently.
A New Paradigm for Catalyst Development
In reshaping the landscape of single-atom catalysis, this research not only deepens our understanding of electronic interactions at the atomic level but also positions itself as a guiding light for subsequent studies. The insights gleaned are not just academic; they have profound implications for industrial applications, where striving for higher efficiency and reduced costs are essential. As catalysis continues to evolve, its role in sustainability and energy transformation becomes increasingly significant, making the quest for optimized single-atom catalysts crucial.
Prof. Wensheng and his team have ignited a new trajectory in catalyst development, leaving the scientific community pondering the endless possibilities that meticulous research and innovative methodologies can unlock in the field of catalysis.
Leave a Reply